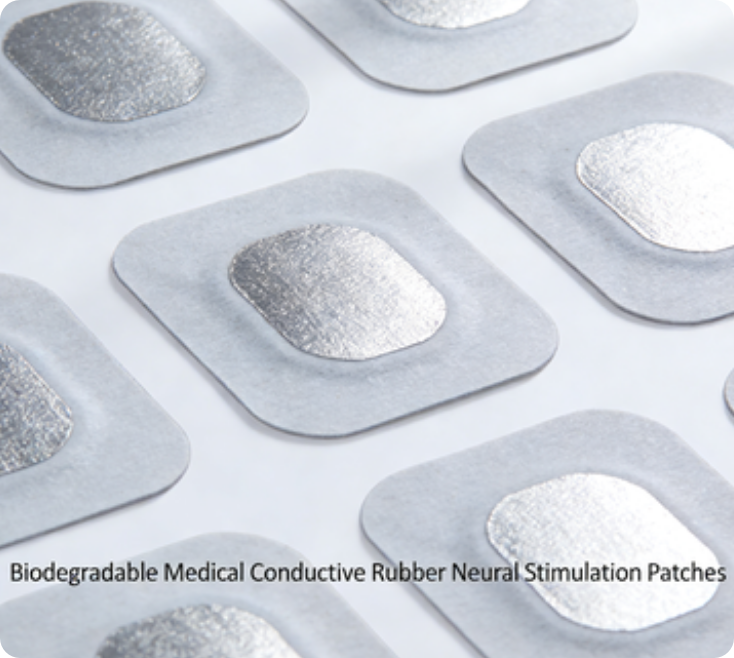
Electrically conductive rubber products for biodegradable medical conductive rubber neural stimulation patches
I. Urgent Need to Solve Clinical Pain Points: Traditional Solutions Hit a Bottleneck
II. Integration of Three Core Innovations: Setting a New Benchmark for Temporary Medical Implants
-
Guaranteed biocompatibility
Using medical‑grade biodegradable polymer substrates and biofriendly conductive fillers, it has passed rigorous biosafety testing. It causes no rejection with human tissue and no irritation when attached to wounds, providing a safe foundation for temporary in vivo treatment.
-
Stable and non‑attenuating conductivity
Featuring an innovative conductive pathway design, it maintains stable electrical conductivity for 1–2 weeks even in a bodily fluid environment, ensuring therapeutic precision for neural stimulation and pain management to meet clinical treatment cycle requirements.
-
Controllable degradation without residue
After the treatment cycle, the patch naturally degrades into non‑toxic small‑molecule metabolites (such as lactic acid and water) in the body, which are excreted through normal human metabolism. No secondary surgery is required for removal, completely eliminating trauma and infection risks associated with extraction.
III. Precise Scenario Application: Empowering a New Experience in Postoperative Rehabilitation
-
Spinal cord injury rehabilitation
Within 1–2 weeks after surgery, stable electrical stimulation promotes neural function repair. The patch degrades naturally alongside the rehabilitation process, avoiding additional stimulation to the spinal cord from secondary surgery.
-
Postoperative pain management
It acts precisely on pain targets, continuously releasing mild electrical signals to relieve pain without long‑term foreign body retention, reducing risks of infection and tissue adhesion.
-
Other temporary electrical stimulation needs
Such as peripheral nerve injury repair and postoperative muscle function recovery, adapting to various short‑term treatment scenarios after minimally invasive surgery.
IV. Highlighting Core Differences: Leading Industrial Technological Innovation
-
Breaking material limitations
Abandoning non‑degradable conductive rubber, it adopts biodegradable substrates, balancing conductive stability and in vivo safety.
-
Simplifying treatment procedures
Eliminating the need for secondary removal, it reduces patient suffering and medical costs while improving clinical treatment efficiency.
-
Reducing safety risks
Fundamentally avoiding complications such as infection and rejection caused by foreign body retention, safeguarding the quality of postoperative rehabilitation.